Solved 1. A cup of hot 50°C water is poured onto 35g of ice

Answer to Solved 1. A cup of hot 50°C water is poured onto 35g of ice

Solved 1. A cup of hot 50°C water is poured onto 35g of ice

8.2: Calorimetry (Problems) - Chemistry LibreTexts

Clandestine Info, PDF, Water

19 top Vegan Hair Care Products ideas in 2024

Chem dems - www-personal.edfac.usyd.edu.au
What mass of ice is needed to cool 60g of water from 45°C to 20°C? - Quora

Modernist cuisine at Home by Aaron Y - Issuu

Solved 1. A cup of hot 50°C water is poured onto 35g of ice
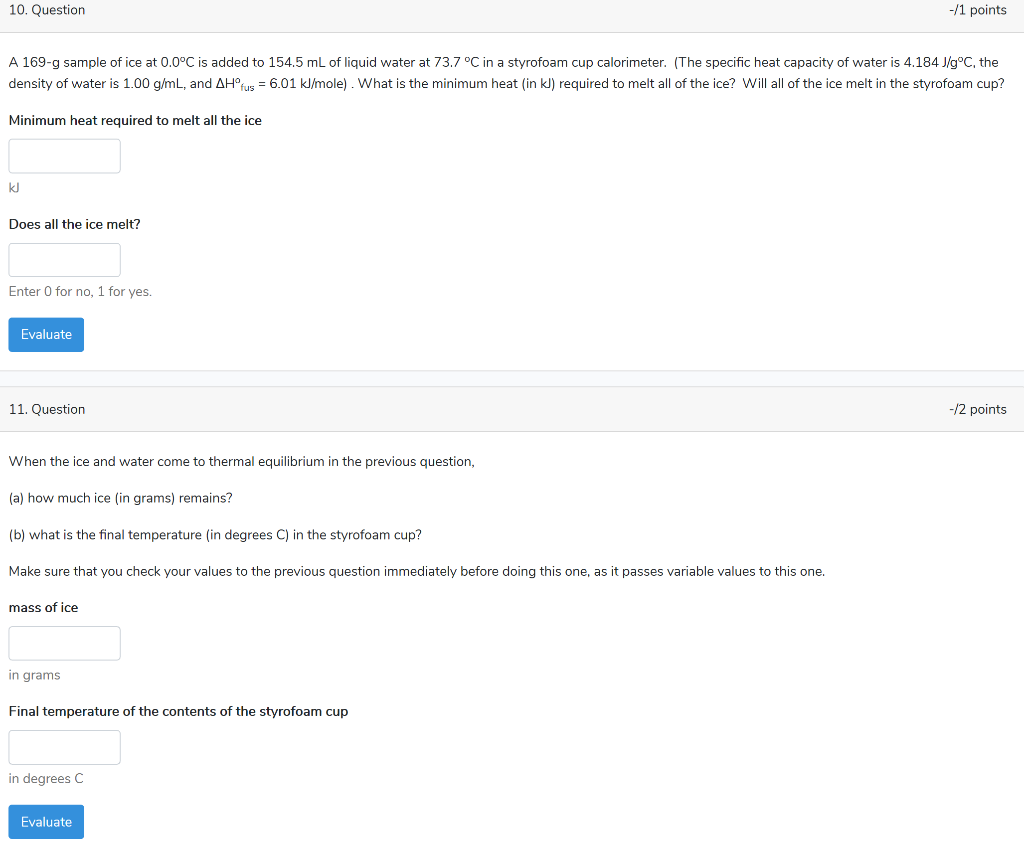
Solved 10. Question -/1 points A 169-g sample of ice at

Calaméo - Guide d'achat 2021-2022 EN

First for Women January 16, 2023 (Digital)
SOLVED: A cup of hot 50°C water is poured onto 35g of ice at 0°C until the entire system ends up at 20°C. How much energy is needed to melt the 35g
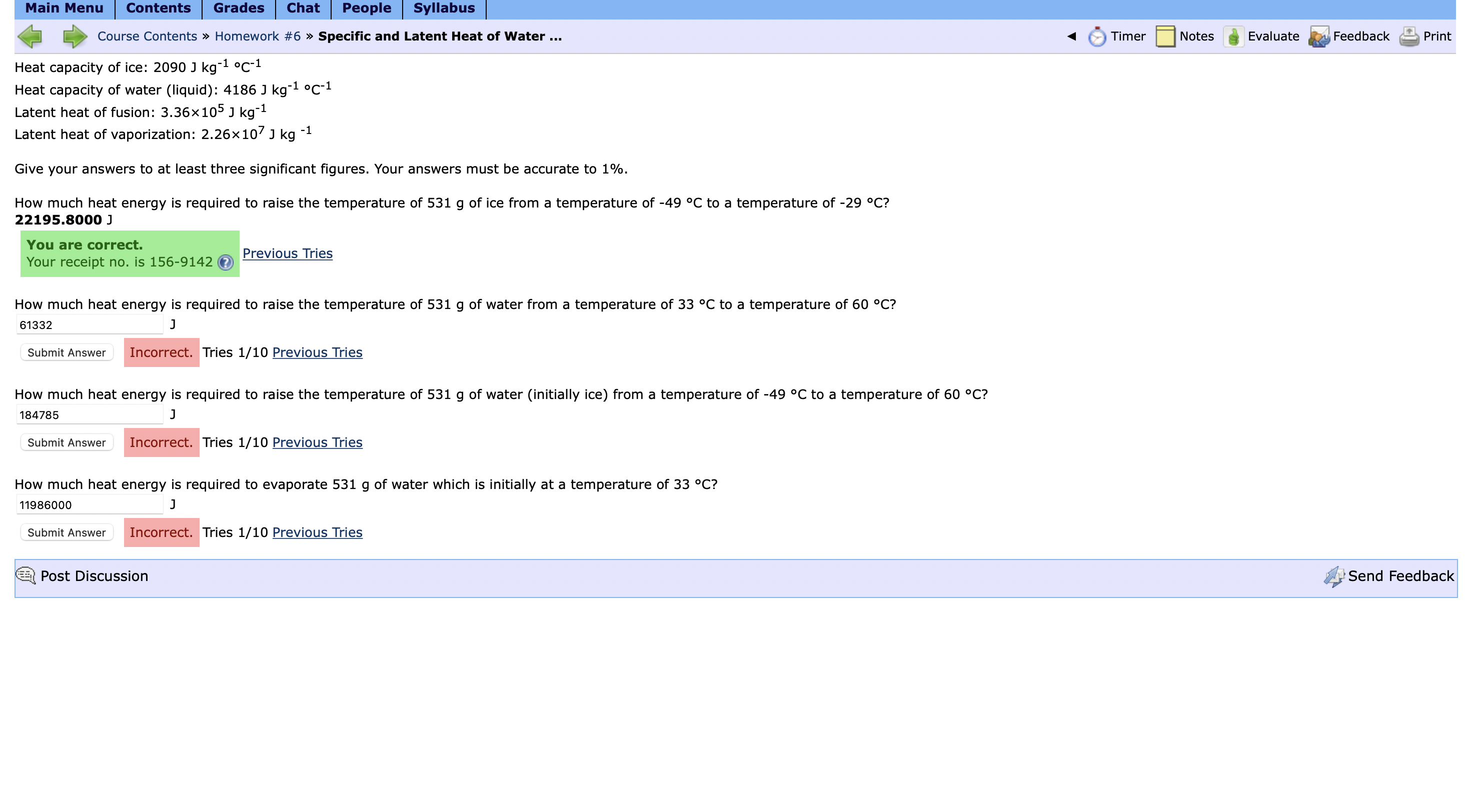
Solved Heat capacity of ice: 2090Jkg-1°C-1Heat capacity of

An ice cube whose mass is 50 g is taken from a refrigerator where its temperature was`-10^C`. If

SOLVED: A 35 g block of ice at -14°C is dropped into a calorimeter (of negligible heat capacity) containing 400 g of water at 0°C. When the system reaches equilibrium, how much





)

